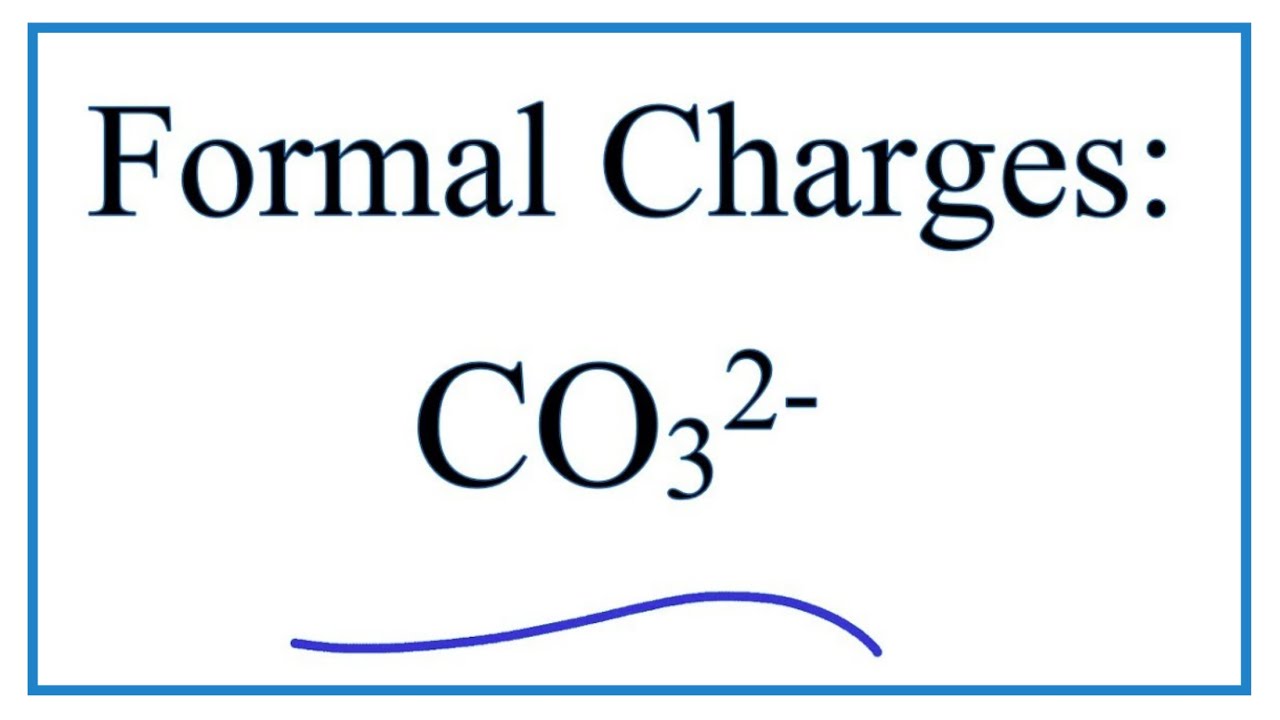
During chemical reactions, it is common to have charge reactant, intermediates, and/or products. Organic molecules can also have positive or negative charges associated with them. In the beginning, it can be helpful to physically add the lone pair electrons.įor organic chemistry, the common bonding patterns of carbon, oxygen, and nitrogen have useful applications when evaluating chemical structures and reactivity. Since the lone pair electrons are often NOT shown in chemical structures, it is important to mentally add the lone pairs. Unshared electrons are also called ‘Lone Pairs’ and are shown as ‘:’ Double and triple bonds can also be communicated with lines as shown below.Ģ shared electrons form a single bond shown as ‘:’ or ‘–‘Ĥ shared electrons form a double bond shown as ‘::’ or ‘=’Ħ shared electrons form at triple bond shown as ‘:::’ or Lone pair (unshared) electrons are still shown as individual electrons. Line representations are only used for shared electrons. While it can be helpful initially to write the individual shared electrons, this approach quickly becomes awkward.Ī single line is used to represent one pair of shared electrons. Lewis structures, also known as Lewis-dot diagrams, show the bonding relationship between atoms of a molecule and the lone pairs of electrons in a molecule. 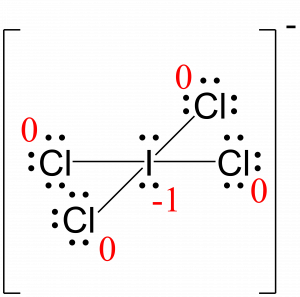
Note: The review of general chemistry in sections 1.3 - 1.6 is integrated into the above Learning Objective for organic chemistry in sections 1.7 and 1.8. 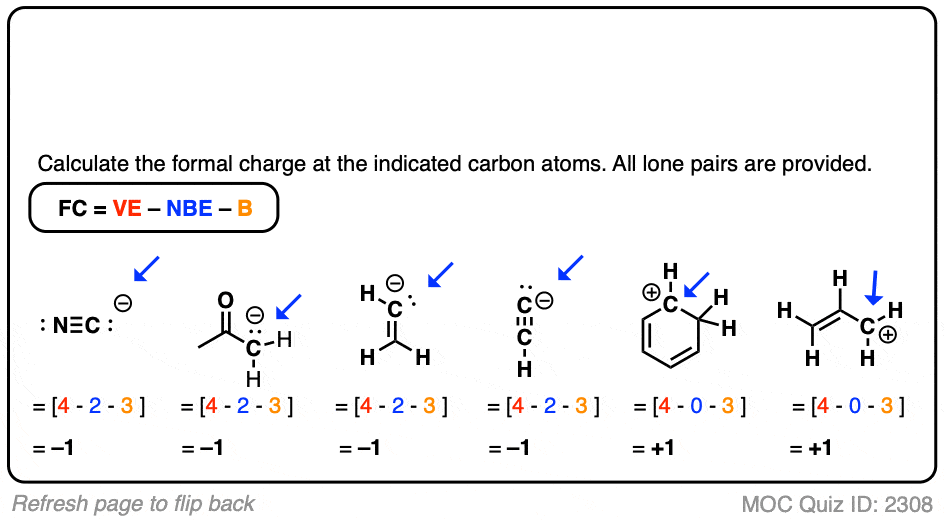
\)ĭraw, interpret, and convert between Lewis (Kekule), Condensed, and Bond-line Structures
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
